Abstract
Theranostics, which is the combination of both therapeutic and diagnostic capacities in one dose, is a promising tool for both clinical application and research. Although there are many chromophores available for optical imaging, their applications are limited due to the photobleaching property or intrinsic toxicity. Curcumin, a natural compound extracted from the rhizome of curcuma longa, is well known thanks to its bio-pharmaceutical activities and strong fluorescence as biocompatible probe for bio-imaging. In this study, we aimed to fabricate a system with dual functions: diagnostic and therapeutic, based on poly(lactide)-tocopheryl polyethylene glycol succinate (PLA-TPGS) micelles co-loaded curcumin (Cur) and paclitaxel (PTX). Two kinds of curcumin nanoparticle (NP) were fabricated and characterized by Fourier transform infrared spectroscopy, field emission scanning electron microscopy and dynamic light scattering methods. The cellular uptake and fluorescent activities of curcumin in these systems were also tested by bioassay studies, and were compared with paclitaxe-oregon. The results showed that (Cur + PTX)-PLA-TPGS NPs is a potential system for cancer theranostics.
Export citation and abstract BibTeX RIS

Original content from this work may be used under the terms of the Creative Commons Attribution 3.0 licence. Any further distribution of this work must maintain attribution to the author(s) and the title of the work, journal citation and DOI.
1. Introduction
The term 'theranostics' is defined as the combination of both therapeutic and diagnostic capacities in one dose. Instead of using separately therapeutic and diagnostic agents for these two purposes, theranostics combines two features into one 'package' which is potential for avoiding the undesirable differences in biodistribution and selectivity between two agents resulting in the more effective diagnostic and treatment [1]. In addition, theranostics also facilitates the process for researching and accessing the potential of new drug molecules or new delivery systems in all stages of drug development process from in vitro, in vivo to clinical research.
For visualizing, many imaging methods such as magnetic resonance imaging, positron emission tomography and optical imaging are applied in which optical imaging has advantage of simplicity and cost-effectiveness [2]. Although there are many chromophores available for optical imaging such as organic dyes or quantum dots, their applications are limited due to the photobleaching [3] or intrinsic toxicity [4]. Therefore, seeking new molecules which possess good fluorescent property and non-toxicity is of interest.
Curcumin, a natural yellow compound extracted from the rhizome of curcuma longa, is well known thanks to it wide range of bio-pharmaceutical activities against various types of disease such as type II diabetes, rheumatoid arthritis, alzheimer's disease and many kinds of cancer including gastrointestinal, melanoma, breast, lung, head and neck, neurological and sarcoma. In addition, curcumin was proven as a friendly-therapeutic agent to healthy cells [5]. Beside its bio-pharmaceutical activities, curcumin also exhibits strong fluorescence as biocompatible probe for bio-imaging [6]. Garcial-Alloza et al [7] used curcumin as fluorescent agent for ex vivo and in vivo monitoring the structural changes of amyloid deposits in alzheimer treatment. However, the poor water solubility of curcumin limited its application for both diagnostics and treatment. To solve this problem, various types of nanocarriers were researched and developed. These nanocarriers improved the curcumin's solubility and significantly increased the photostability which was one of the limitations of organic dyes [8, 9]. Very recently, Nagahama et al [10] fabricated nanoparticle of dextran-curcumin conjugate which was effectively delivery into cancer cell and exhibited strong fluorescence available for live-cell imaging.
Cancer is more and more increasing and becoming a huge challenge for human kind. Along with discovering new anti-cancer drugs and better diagnostic methods, developing novel delivery nanosystems plays an important role on improving the efficiency of current therapeutic and diagnostic agents through prolonging circulation time and selective targeting to cancer cell and tumor [11]. Many kinds of nanosystem have been developed such as liposome, dendrimer, virus-based nanoparticle, inorganic nanoparticle, solid lipid nanoparticle and polymeric nanoparticle [12]. Among them polymeric micelle composed by amphiphilic copolymer molecules has attracted great deal of attention thanks to its small size, high drug loading capacity and excellent stability [13].
In previous works [14–16] we have used polymeric micelles composed by poly (lactide)-tocopheryl polyethylene glycol succinate (PLA-TPGS) copolymer as a nanocarrier for loading and delivering hydrophobic drugs such as paclitaxel and curcumin serving for chemotherapy. Up to date, to the best of our knowledge, there was a few publications mentioning the use of curcumin as fluorescent probe for monitoring the delivery and biodistribution of drug delivery systems. In this article, in order to fabricate a system with dual functions: diagnostic and therapeutic, curcumin was co-loaded with paclitaxel in the hydrophobic core of the PLA-TPGS micelles. Uptake of the system into MCF7 cell and MCF7 spheroid was monitored based on the fluorescent signal of curcumin under confocal fluorescence microscopy. The results showed curcumin as a potential fluorescent probe for monitoring the delivery and biodistribution of the drug delivery system.
2. Material and methods
2.1. Materials
PLA-TPGS copolymer was obtained from Laboratory of biomedical nanomaterials, Institute of Materials Science, Vietnam Academy of Science and Technology. Curcumin and paclitaxel were purchased from Sigma-Aldrich. Solvents (dichloromethane, ethanol) were purchased from Merck (Germany). Distilled water was used for all experiments.
MCF7 breast cell line was obtained from Department of Biology, Hanoi University of Science. Solvents and chemical for bioassays were purchased from Invitrogen.
2.2. Methods
2.2.1. Preparation of curcumin nanoparticles
Two kinds of curcumin nanoparticles, curcumin loaded PLA-TPGS nanoparticles (Cur-PLA-TPGS NPs) and (curcumin + paclitaxel) co-loaded PLA-TPGS nanoparticles ((Cur + PTX)-PLA-TPGS NPs), were prepared by the emulsification solvent evaporation method. In brief, 100 ml aqueous solution of copolymer PLA-TPGS was prepared by adding 200 mg of PLA-TPGS into 100 ml distilled water and stirring for 4 h. Next, curcumin (10 mg) dissolved in 20 ml dichloromethane or mixture of curcumin (10 mg) and paclitaxel (5 mg) dissolved in 20 ml dichloromethane was added dropwise to 50 ml prepared PLA-TPGS solution in a ground bottom flask under vigorously stirring. The flask was closed and the systems were stirred for 24 h. After that, the solvent was evaporated. Then, the obtained mixtures were centrifuged at 5600 rpm in 10 min. The transparent solutions were collected, parts of them were lyophilized and the remains were stored at 4 °C.
2.2.2. Characterization methods
Molecular structure of Cur-PLA-TPGS NPs and (Cur + PTX)-PLA-TPGS NPs was characterized by Fourier transform infrared spectroscopy (FTIR, Shimadzu spectrophotometer) using KBr pellets in the wave number region of 400–4000 cm−1. Their morphology was investigated by field emission scanning electron microscopy on a Hitachi S-4800 system. Size distribution was measured by dynamic light scattering method.
Drug loading content (LC) was determined with a UV–vis spectrophotometer. Calibration curve was obtained with ethanolic solutions of curcumin and paclitaxel. Dry samples of nanoparticles (5 mg) were immersed in 10 ml ethanol and stirred for 6 h in closed flask. The obtained ethanolic solutions of drugs were measured at 230 nm for paclitaxel and 432 nm for curcumin. The LC was calculated based on the following equation
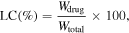
where Wdrug is the weight of loaded drug, Wtotal is the total weight of polymers.
2.2.3. Cell and spheroid culture
MCF7 cells were activated and cultured under atmosphere of 5% CO2 and 95% air at 37 °C. Cell culture medium was refreshed every 2 days to ensure sufficient nutrients and remove death cells.
MCF7 spheroids were prepared by adding 5000 cells in 20 μl cell medium into each well of 96-well plate, previously contained 60 μl agarose 1.5% and 180 μl RPMI 1640 medium (Gibco) and incubated under atmosphere of 5% CO2 and 95% air at 37 °C. The culture medium was refreshed every 2 days.
2.2.4. In vitro uptake of curcumin nanoparticles into MCF7 cell and MCF7 tumor spheroid
MCF7 cells were exposed to pure curcumin, curcumin nanoparticles ((Cur-PLA-TPGS NPs) and (Cur + PTX)-PLA-TPGS NPs) for 24 h with the concentration of 0.4 μg ml−1 curcumin in all forms and 0.2 μg ml−1 paclitaxel. Cells were washed three times with phosphate buffered saline (PBS, Invitrogen) and continued to stain with Hoechst (Invitrogen) for 15 min and then washed three times with PBS. Cell images were taken by laser scanning confocal microscope.
Mature spheroids were exposed to pure curcumin, curcumin nanoparticles, and paclitaxel-oregon for 24 h with the same above-mentioned concentration of curcumin and paclitaxel. The spheroids were washed three times with PBS. Spheroid images were taken by LMSC.
3. Results and discussion
3.1. Characteristics of nanoparticles
The LC of Cur-PLA-TPGS NPs was about 85%, while for (Cur + PTX)-PLA-TPGS NPs, the curcumin LC slightly decreased to 76% and the paclitaxel LC was about 82%.
The interaction between drugs and copolymer was investigated by FTIR spectroscopy. Figure 1 shows the FTIR spectrum of PLA-TPGS, pure curcumin, pure paclitaxel, Cur-PLA-TPGS NPs and (Cur + PTX)-PLA-TPGS. The characteristic vibration of PLA-TPGS at 1756 cm−1 which is attributed to the carbonyl group (C=O) stretching [16] was shifted to 1746 cm−1 and characteristic vibrations of pure curcumin at 1620 and 1285 cm−1 were shifted to 1605 cm−1 and 1279 cm−1 in the spectrum of Cur-PLA-TPGS NPs, respectively. In the spectrum of (Cur + PTX)-PLA-TPGS NPs, there were clear changes compared to FTIR spectrum of pure paclitaxel, pure curcumin and PLA-TPGS. The carbonyl group (C=O) stretching of PLA-TPGS (1756 cm−1) and curcumin (1620 cm−1) were shifted to 1740 cm−1 and 1600 cm−1, respectively, while C=O stretching of paclitaxel (1720 cm−1) was disappeared. It could be overlapped by vibrations of carbonyl groups of curcumin and PLA-TPGS. The peaks at 1630 cm−1 and 1465 cm−1 were assigned to C–C stretching of paclitaxel (1650 cm−1 in pure paclitaxel) and C=C olefienic stretching of curcumin (1502 cm−1 in pure curcumin). Furthermore, there were significant changes in the region of 400–1330 cm−1. All of above analyses demonstrated the success in loading curcumin and paclitaxel into PLA-TPGS micelles.
Figure 1. FTIR spectra of (1) PLA-TPGS, (2) curcumin (Cur), (3) paclitaxel (PTX), (4) Cur-PLA-TPGS NPs and (5) (Cur + PTX)-PLA-TPGS NPs.
Download figure:
Standard image High-resolution image3.2. Morphology and size distribution
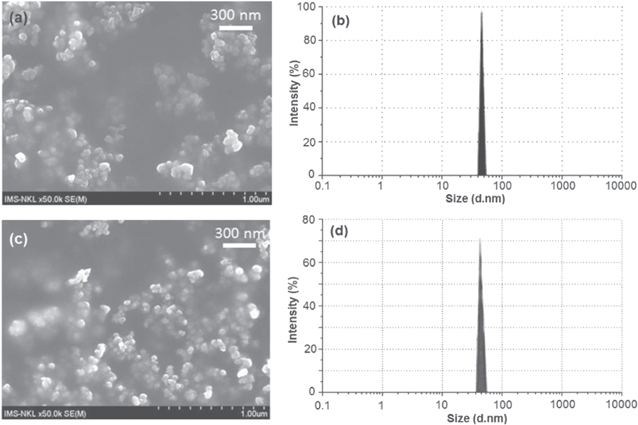
It is well-known that nanoparticles with the small size (below 200 nm) can exist longer in the blood circularly system and easily accumulate into tumor site via the enhanced permeability and retention effect [17]. Therefore, for both diagnostic and treatment purposes, the delivery system must be small enough to maximize the efficiency and avoid being filtered by interendothelial cell slits at the spleen and then removed by phagocytic. In this research, (Cur + PTX)-PLA-TPGS NPs have very small size of about 50 nm and narrow size distribution (figure 2). This will enhance the uptake of the nanoparticle into the cell or tumor resulting improving the efficiency of system.
Figure 2. FESEM images and size distribution of Cur-PLA-TPGS NPs (a), (b) and (Cur + PTX)-PLA-TPGS NPs (c), (d).
Download figure:
Standard image High-resolution image3.3. Fluorescence spectra
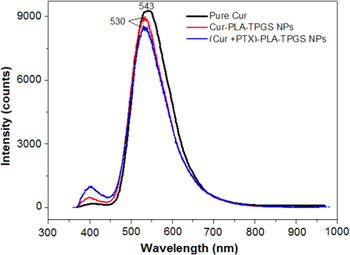
Our previous publication showed that Cur-PLA-TPGS NPs exhibit good fluorescent emission efficiency [16]. In this study we used fluorescence spectra to access whether the emission of curcumin changes in the presence of paclitaxel (curcumin and paclitaxel were co-loaded into the hydrophobic core of PLA-TPGS micelles). Emission peak of curcumin in ethanol at 543 nm was shifted to 530 nm after loaded into PLA-TPGS micelles. This change may be due to the hydrophobic interaction of curcumin with the hydrophobic core of PLA-TPGS micelles. The emission peak of curcumin in (Cur + PTX)-PLA-TPGS NPs was not shifted and slightly decreased in fluorescence intensity (figure 3). This seems that paclitaxel does not induce large impact to the fluorescence emission of curcumin.
Figure 3. Fluorescence spectra of curcumin, Cur-PLA-TPGS NPs and (Cur + PTX)-PLA-TPGS NPs.
Download figure:
Standard image High-resolution image3.4. Cellular uptake of curcumin nanoparticles
The potential of (Cur + PTX)-PLA-TPGS NPs as the system available for cancer theranostics was investigated through its uptake into MCF7 cells and MCF7 spheroid. The distribution of nanoparticle inside cells and spheroid was observed on fluorescent images obtained by confocal fluorescence microscopy when curcumin was excited at wavelength of 488 nm. In the case of MCF7 cell, the distinct difference in cellular uptake of curcumin nanoparticles (Cur-PLA-TPGS NPs and (Cur + PTX)-PLA-TPGS NPs) and pure curcumin through the fluorescence intensity (figure 4) was clearly obtained. Weak green fluorescence intensity was observed for the case of pure curcumin indicating the low cellular uptake of pure curcumin into the cancer cells. In contrast, for curcumin nanoparticles, strong green fluorescence intensity was observed indicating high cellular uptake of curcumin nanoparticles into the cancer cells. Curcumin nanoparticles mainly located in the cytoplasm of cancer cells in monolayer culture.
Figure 4. Cellular uptake of different forms of curcumin after 24 h of treatment: (a) control MCF7 cells, (b) pure curcumin-treated MCF7 cells [18], (c) Cur-PLA-TPGS NPs-treated cells [18], (d) (Cur + PTX)-PLA-TPGS NPs-treated cells (hoechst-blue light, curcumin-green light).
Download figure:
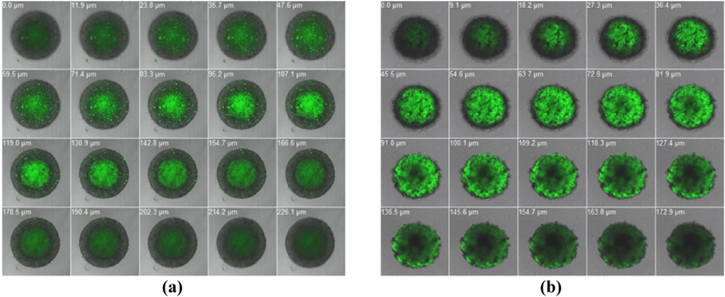
Standard image High-resolution imageFurther experiment was performed on the MCF7 spheroids. Multicellular spheroids are spherical aggregates of tumor cells that reflect many properties of solid tumor. By incubating spheroids in a medium that contained an anticancer drug, it is possible to examine the kinetics of drug penetration in histological sections. In our experiment, the curcumin nanoparticles also exhibited better uptake into the spheroids compared to that of pure curcumin (figure 5). Curcumin was presented mostly in outer layer cells meanwhile curcumin nanoparticles localized not only in outer layer but also in the middle layer, especially (Cur + PTX)-PLA-TPGS NPs successfully came into the necrotic core.
Figure 5. The absorption of different forms of curcumin in MCF7 multicellular tumor spheroids after 24 h of incubation: (a) control spheroid, (b) pure curcumin-treated spheroid, (c) Cur-PLA-TPGS NPs-treated spheroid and (d) (Cur + PTX)-PLA-TPGS NPs-treated spheroid.
Download figure:
Standard image High-resolution imageFigure 6 showed the different layers of spheroids after 24 h incubated with paclitaxel-oregon and (Cur + PTX)-PLA-TPGS NPs. In comparison with paclitaxel-oregon, which is normally used for monitoring biodistribution of paclitaxel, the fluorescence signal of (Cur + PTX)-PLA-TPGS NPs had not only similar intensity but also wider distribution than that of commercial product. After 24 h of treatment, paclitaxel-oregon mostly concentrated in the necrotic core of tumor spheroid, in the meantime (Cur + PTX)-PLA-TPGS NPs presented in the whole spheroid. Moreover, the nanoparticles showed the most efficient effect on the inhibition of tumor spheroid growth with smallest size compare with the controls (figures 5 and 6). This evidence indicated the high potential of (Cur + PTX)-PLA-TPGS NPs for cancer theranostics. Figure 7 shows the diameter values of spheroid treated with paclitaxel-oregon.
Figure 6. The biodistribution of Paclitaxel-oregon and (Cur + PTX)-PLA-TPGS NPs in diferent layers of MCF7 tumor spheroids: (a) PTX-oregon concentrated mostly in the necrotic core meanwhile (Cur + PTX)-PLA-TPGS NPs, (b) distributed from outer layer to necrotic core of spheroid. The upper-left number indicates the depth of spheroid layer.
Download figure:
Standard image High-resolution imageFigure 7. The diameter values of spheroid treated with paclitaxel-oregon.
Download figure:
Standard image High-resolution imageRecently, many researches had been performed on tumor cells and tumor spheroids to evaluate the relationship between the particles size and their penetration. In 2013 Huo et al [19]found that for Au@tiopronin nanoparticles, more uptake was observed for the 50 nm nanoparticles compared to that of the 100 nm ones. This was also in agreement with the results of Chithrani et al [20]. Moreover, the penetration into spheroid tumor of the 50 nm particles increased in both the depth and the quantity with prolonging incubation time from 3 to 24 h. However, the 100 nm particles were hindered outside of the tumor spheroid [19]. All these data indicated that the size of the nanomedicines critically affects the penetration and efficacy of the drug in tumors.
4. Conclusion
In this study curcumin nanoparticles (Cur-PLA-TPGS NPs, (Cur + PTX)-PLA-TPGS NPs) were fabricated with the aim of accessing fluorescent properties of curcumin for cancer theranostics. The results showed that curcumin nanoparticles exhibit better uptake to MCF7 cells and MCF7 spheroid than that of pure curcumin. The co-loading of curcumin and paclitaxel into PLA-TPGS micelles did not induce much change to the fluorescent property of curcumin. The comparison of fluorescent property of paclitaxel-oregon and (Cur + PTX)-PLA-TPGS NPs on MCF7 spheroid showed that (Cur + PTX)-PLA-TPGS NPs distributed from outer layer to necrotic core of spheroid compared to the only necrotic core-distribution of paclitaxel-oregon. This is the strong evidence of potential of (Cur + PTX)-PLA-TPGS NPs for cancer theranostics.
Acknowledgments
This work was financially supported by the Vietnam Academy of Science and Technology under Grant No.VAST03.04/16-17 (HPT) and the Vietnam National University, Hanoi code: QGTĐ 10.28 (HTMN).